|
8/12/2023 0 Comments Keynote 177 colorectalThe ORR was 43.8% with pembrolizumab, which consisted of an 11.1% complete response (CR) rate, 32.7% partial response (PR) rate, and 20.9% stable disease (SD) rate. 
Results also showed that the 12-month PFS rates were 55% in the pembrolizumab arm and 37% in the chemotherapy arm, and the 24-month PFS rates were 48.3% and 18.6%, respectively. Patients’ baseline and demographic characteristics were generally well balanced between arms. All responses were evaluated according to RECIST v1.1 criteria by blinded independent central review. PFS and overall survival served as the coprimary endpoints of the study key secondary end points included overall response rate (ORR) and safety. Treatment was continued for up to 35 cycles in the pembrolizumab arm, until progressive disease, unacceptable toxicity, or patient/physician decision to withdraw. Patients in the chemotherapy arm were allowed to cross over to the pembrolizumab arm at the time of disease progression. Patients were randomized 1:1 to 200 mg of pembrolizumab every 3 weeks for up to 2 years or investigator’s choice of standard chemotherapy, including intravenous (IV) mFOLFOX6 every 2 weeks mFOLFOX6 plus bevacizumab mFOLFOX6 plus cetuximab (400 mg/m 2 IV, then 250 mg/m 2 weekly in each 2-week cycle) FOLFIRI (leucovorin, 5-FU, and irinotecan) FOLFIRI plus bevacizumab (5 mg/kg IV on day 1 of each 2-week cycle) or FOLFIRI plus cetuximab (400 mg/m 2 IV, then 250 mg/m2 weekly in each 2-week cycle). To be eligible for enrollment, patients had to have confirmed MSI-H/dMMR stage IV CRC, an ECOG performance status of 0 or 1, and measurable disease. KEYNOTE-177 included 307 patients with treatment-naïve MSI-H/dMMR mCRC. The approval was based on data from 149 patients with MSI-H/dMMR cancers enrolled across 5 single-arm clinical trials, in which pembrolizumab demonstrated durable responses and prolonged survival. In May 2017, the FDA granted an accelerated approval to pembrolizumab for the treatment of adult and pediatric patients with unresectable or metastatic, MSI-H/dMMR solid tumors that have progressed after prior treatment and who have no satisfactory alternative treatment options, as well as for patients with MSI-H/dMMR CRC following progression on a fluoropyrimidine, oxaliplatin, and irinotecan. In prior phase 2 trials, MSI-H status demonstrated predictive clinical benefit for pembrolizumab, as well as other PD-1 inhibitors, in patients with chemotherapy-refractory mCRC. Patients with metastatic MSI-H/dMMR tumors are less responsive to conventional chemotherapy and are in need of alternative treatment options. This randomized study demonstrates a huge benefit in the first-line with pembrolizumab and should be the new standard of care.”Īpproximately 5% of patients with mCRC have microsatellite instability, categorized by a high number of mutations in the tumor and decreased survival. 2 “Pembrolizumab works in non-randomized studies in this group of patients with advanced disease. “These long-awaited trial results will change clinical practice,” said lead author Thierry André, MD, of the Sorbonne Université and Hôpital Saint Antoine in Paris, in a press briefing ahead of the virtual conference. 
1Īt the time of data cutoff, in February 2020, and at a median follow-up of 32.4 months, the median PFS was 16.5 months (95% CI, 5.4-32.4) with pembrolizumab (n = 153) versus 8.2 months (95% CI, 6.1-10.2) with standard chemotherapy with or without bevacizumab (Avastin) or cetuximab (Erbitux n = 154) in patients with MSI-H/dMMR mCRC, leading to a 40% reduction in the risk of disease progression or death (HR, 0.60 95% CI, 0.45-0.80 P =. 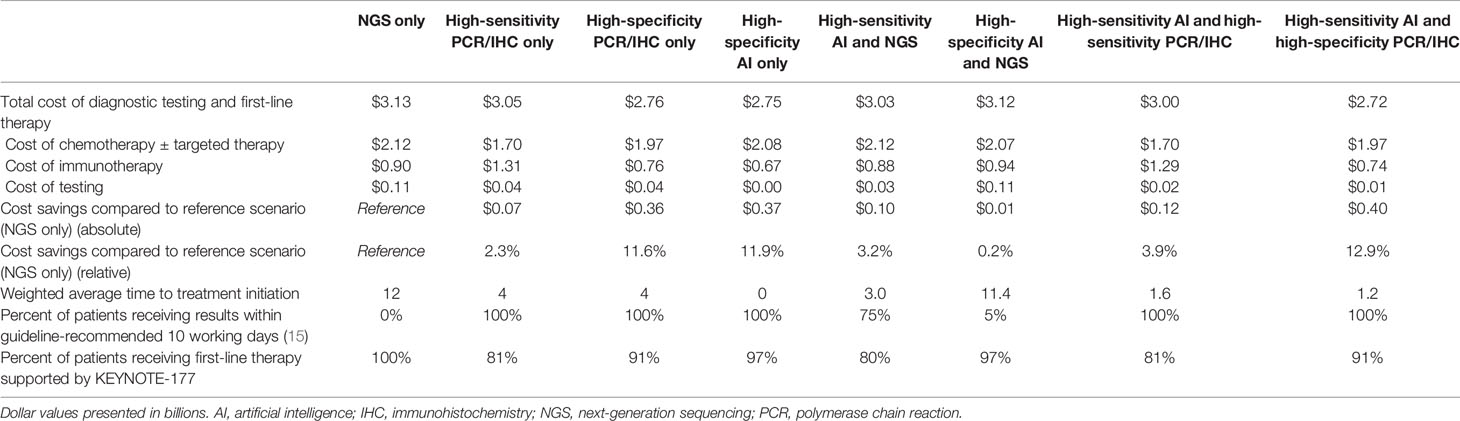
Pembrolizumab (Keytruda) doubled progression-free survival (PFS) compared with chemotherapy in patients with newly diagnosed microsatellite instability-high (MSI-H)/mismatch repair deficient (dMMR) metastatic colorectal cancer (mCRC), according to findings from an interim analysis of the phase 3 KEYNOTE-177 trial (NCT02563002) that were presented ahead of the 2020 ASCO Virtual Scientific Program.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
